Canada has spent years sitting in an awkward position on CBD.
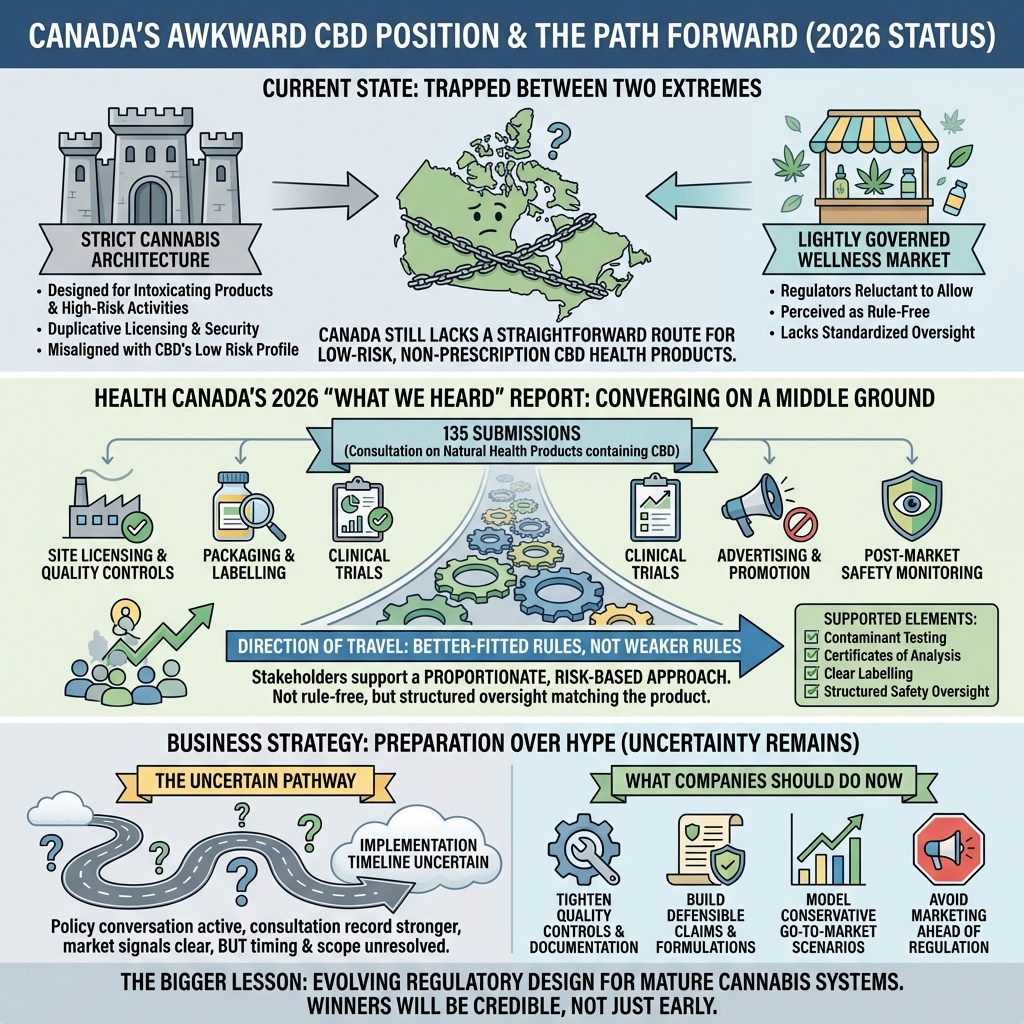
The country has a federally legal cannabis framework, a regulated industrial hemp system, sophisticated testing and quality expectations, and clear consumer interest in cannabidiol products. Yet when it comes to low-risk, non-prescription health products containing CBD, Canada still does not offer a straightforward route to market.
That is why Health Canada’s February 2026 “What we heard” report matters so much.
The department said it received 135 submissions in response to its 2025 discussion paper on a possible pathway for natural health products containing cannabidiol. The consultation covered five major areas: site licensing and quality controls, packaging and labelling, clinical trials, advertising and promotion, and post-market safety monitoring.
The feedback was useful not because it finalized a pathway, but because it made the direction of travel easier to read.
Why Canada still does not have a clean CBD route to market
For years, Canadian CBD policy has been trapped between two extremes.
On one side sits the existing cannabis architecture, which applies strict controls that were designed for intoxicating products and higher-risk activities. On the other sits the idea of a lightly governed wellness market, which regulators have been understandably reluctant to allow.
The consultation responses point toward a more credible middle ground.
Across many sections, respondents pushed for a proportionate, risk-based approach that treats low-risk CBD differently from intoxicating cannabis products. Most did not support requiring a cannabis drug licence for low-risk CBD natural health products. Many argued that CBD has low misuse potential and should not be forced through duplicative licensing and security expectations that are misaligned with the product’s actual risk profile.
That does not mean stakeholders were asking for a rule-free market. Quite the opposite. There was broad support for contaminant testing, certificates of analysis, clear labelling, and structured safety oversight.
In other words, the conversation is not about whether CBD should be regulated. It is about whether it should be regulated in a way that matches the product.
What Health Canada’s 2026 consultation feedback really signalled
The most important message from the consultation is that many stakeholders are converging around the idea of better-fitted rules, not weaker rules.
That distinction matters for SEO and for strategy. A lot of public conversation around CBD still falls into a false binary:
- either treat CBD exactly like broader cannabis
- or let it flow into the market like a standard wellness ingredient
The consultation feedback suggests there is room for a more structured, evidence-based middle path.
If that middle path emerges, it will likely still require:
- quality-controlled manufacturing
- contaminant and potency testing
- defensible label claims
- tighter advertising controls than conventional wellness products
- post-market monitoring and clear adverse-event logic
That is why the right business response is not hype. It is preparation.
Why uncertainty still remains in Canada’s CBD pathway
The 2026 story is not “pathway approved.” It is “pathway still under consideration.”
That nuance matters.
In January 2026, StratCann reported that Health Canada’s latest Forward Regulatory Plan no longer explicitly referenced the earlier proposal to regulate CBD products that would not require practitioner oversight, even though the department said it still recognized the importance of the work.
Taken together, these developments create a more realistic picture:
- the policy conversation is active
- the consultation record is now stronger
- the market has signalled what kind of framework it wants
- the route from consultation to implementation is still uncertain in timing and scope
For operators, that means Canada’s CBD market is still a strategic watch area rather than a resolved commercial channel.
What companies should do before a CBD pathway opens
There is still meaningful upside here if Canada eventually creates a fit-for-purpose route for low-risk CBD products. Businesses with strong quality systems, testing discipline, formulation credibility, and regulatory literacy will be better positioned than businesses that wait for final rules before taking the topic seriously.
The smartest companies should use this period to:
- tighten documentation and quality controls
- understand likely labelling and evidence expectations
- model conservative go-to-market scenarios rather than optimistic ones
- build formulations and claims strategies that can survive scrutiny
- avoid marketing ahead of regulation
This is a critical point. In transitional policy environments, the companies that ultimately win are not always the earliest to speak. They are often the earliest to become credible.
The bigger regulatory lesson for cannabis in Canada
The CBD debate is also important because it reveals a wider policy challenge inside mature cannabis markets.
Eventually, governments have to decide whether every cannabinoid-adjacent product should sit inside the same regulatory architecture or whether lower-risk categories deserve proportionate pathways. Canada is now visibly working through that question.
That makes the topic strategically valuable for consultants, manufacturers, and investors alike. It is not just a CBD story. It is a regulatory-design story about how mature cannabis systems evolve.
FAQ: CBD regulation Canada
Is CBD fully legal in Canada as a low-risk wellness product?
No. Canada has legal cannabis and hemp frameworks, but it still does not have a simple, fit-for-purpose route for low-risk non-prescription CBD health products.
What did Health Canada’s 2026 consultation show?
It showed strong support for a more proportionate framework with testing, labelling, and safety controls, but without unnecessarily duplicating high-burden requirements meant for higher-risk cannabis products.
Does this mean a CBD pathway is coming soon?
Not necessarily. The direction is clearer, but the implementation timeline is still uncertain.
What should CBD businesses do now?
Prepare for a compliant market before it fully exists. That means building quality systems, realistic regulatory assumptions, and evidence-backed commercial models.
Canada’s CBD story in 2026 is not a launch story yet. It is a transition story. The pathway is moving, but the companies most likely to benefit will be the ones using the waiting period to become operationally ready.
Sources
- Health Canada, Towards a pathway for health products containing cannabidiol: What we heard report, published February 16, 2026
- Health Canada, Consultation: Towards a pathway for health products containing cannabidiol
- StratCann, Health Canada removes CBD NHP regulation proposal from forward regulatory plan, published January 8, 2026




